FLOWFLEX™
COVID-19 Antigen Home Test
ACON Laboratories, Inc. is the only legal manufacturer of the FDA EUA Flowflex COVID-19 Antigen Home Test
The Flowflex™ COVID-19 Antigen Home Test is all you need to determine your family’s COVID-19 status. Find peace of mind with Flowflex! *Other COVID-19 antigen home tests may require a 2nd test 2-3 days after the first. This test procedure overview does not replace the package insert. Before you begin the test, it is important to read and follow the detailed instructions in the package insert.
Get Back to Sharing Special Moments
with Family and Friends
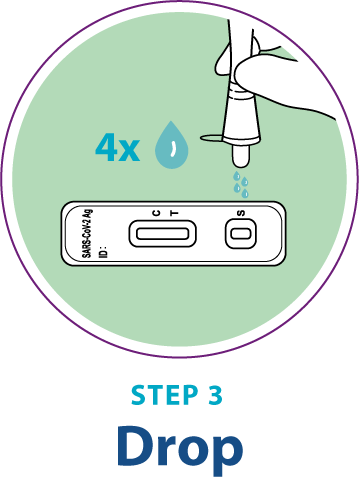
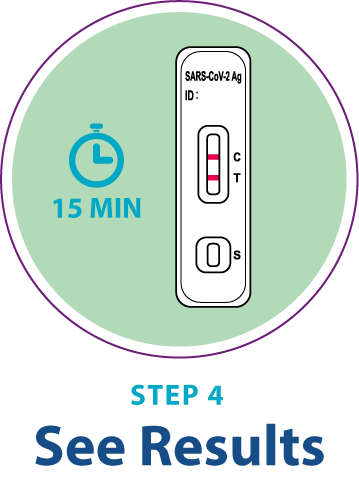
Test Procedure Overview


The Flowflex™ COVID-19 Antigen Home Test may be purchased at the selected retail drug stores and online: For employee testing solutions, click here. To locate an authorized US distributor, please click the button.
Where Can I Buy It?

Looking for a Distributor?
This product has not been FDA cleared or approved but has been authorized by FDA under an EUA.
This product has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens.
The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of IVDs for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated, or authorization is revoked sooner.
For more information on EUAs please visit: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization
For the most up to date information on COVID-19, please visit: www.cdc.gov/COVID19